If a Dialysis Bag Filled With Water
The bags initial mass is 20 g. The bag decreases in weight B.
Bag will gain water so it will weigh less because the water will move from an area of higher solute concentration.

. The two substances that entered the dialysis bag were the iodine solution and water. Check all that are true. Adverse patient outcomes including outbreaks associated with water exposure in dialysis settings have resulted from patient exposure to water via a variety of pathways.
Then I would reweigh the bag. Glucose Starch Solution 15 mL. During an average week of hemodialysis a patient can be exposed to 300-600 liters of water providing multiple opportunities for potential patient exposure to waterborne pathogens.
I would then place the bag in a beaker of water and let it sit for a day. Dialysis bags model the selective permeability of cell membranes and allow very small molecules ions and water to pass through according to the. Solved expert answers for Essentials of Biology 3rd Edition by Sylvia Mader Michael Windelspecht.
A dialysis bag is filled with distilled water and then placed in a sucrose solution. A hypertonic solution is the type of solution which has a higher concentration of solutes on the outside of the cell than on the inside of a cell. If a dialysis bag filled with water is placed in a starch solution what do you predict will happen to the weight of the bag over time.
Water with several drops of iodine added to it until it was visibly yellow-amber was added to a 400ml beaker. The dialysis tubing was clipped to form a bag so that glucose and starch was fed into the bag through the other end and was also clipped to avoid the seeping of the solution. The bags were then placed in separate beakers of distilled water.
This would cause the water to flow out of the bag and into the beaker. Instant access with 247 expert assistance. Iodine turns starch dark blue which happened to definitely be inside the bag.
The bag does not change in weight 44. Next we placed the dialysis tube into a beaker filled with distilled water. We then filled this tube with a starch solution and sealed the other end the same way.
If you want water to flow out of a dialysis bag filled with a 50 sucrose solution what would the minimum concentration of the beaker solution need to be. ANSWER 0 Sexem2001 ANSWERS. Bag C was filled with a 10mL 25 sucrose solution and Bag D was filled with a 10mL 50 sucrose solution.
If a dialysis bag filled with water is placed in a glucose solution what is the result to the weight of the bag over time. Placing red blood cells in a solution less than 09 NaCl will have what impact on these cells. C Water will move out of the bag and into the beaker by osmosis.
Water is the solvent inside the bag and outside in the beaker. Dip a glucose test strip into the water in the cup for 1-2 seconds. 3A dialysis bag is filled with 3 sucrose solution and placed in a beaker contains 30 sucrose solution.
Wait 2-3 minutes to observe any color change on the strip. Calculate the percent change of mass showing your calculations here-18g 20g -2-2 20 -1-1 x 100 -10 6. Run the test strip along the edge of the cup to remove any excess liquid.
The bag increases in weight C. If a dialysis bag filled with water is placed in a starch solution. The sucrose solution in the beaker would have been hypotonic to the distilled water in the bag.
Fill cup with distilled water within 1-2 cm of the top of the cup. A dialysis bag is filled with distilled water and then places in a sucrose solution. Distilled water will move out of the dialysis bag and into the sucrose solution due to osmosis and the fact that the dialysis bag has a hypertonic solution of H2O as compared to the sucrose solution.
The bag will swell and pop. If a dialysis bag filled with water is placed in molasses solution what do you predict will happen to the weight of the bag over time. The dialysis bag will lose water weight in the hypertonic solutions and gain water weight in the hypotonic solutions.
We know iodine diffused into the bag because the substance in the bag turned a dark blue color. Each bag had all the air pushed out of it and the open end was tied. 2 10m pieces of string.
The water will leave the bag by osmosisbecause the. No weight changes will be present in the isotonic solutions. Water Use in Dialysis.
I would test this hypothesis by filling the bag with a solution consisting of water glucose and starch tie it off and weigh it. First we sealed one end of a moist dialysis tube by tying it into a knot. The minimum concentration of the beaker solution would have to be 51 so that the solution must be hypertonic.
What do you predict will happen to the weight of the bag over time. The bags initial mass is 20g and its final mass is 18g. The solution in the beaker is hypotonic to that in the bag with 3 sucrose solution.
RBCs swell in size and. As per the permeability of the dialysis bag it is hypothesized that is it permeable to glucose and sodium but not permeable to. And its final mass is 18 g.
To being the experiment each dialysis tubing was tied off at the bottom and filled with a sucrose solution. Calculate the percent change of mass showing your calculations. A student filled three dialysis bags with different concentrations of a starch solution and one bag was filled with distilled water.
A hypotonic solution. Bag A and Bag B were both filled with a 10mL 1 sucrose solution. The solution in the beaker has to be hypertonic to that in the dialysis bag for water to move from the bag into the beaker.
This is guided by the principles of osmosis where water flows from the hypotonic solution to the hypertonic solution through a semipermeable membrane. Diffusion of Iodine and starch. We also can tell water entered the bag because there was an increase in the size of the bag which proves.
Step 1 of 5.

Biology Or Life Science Lab Diffusion Through A Nonliving Membrane Suitable For Students In Grades 7 12 This Simple Exper Biology Labs Science Cells Biology

Solved Describe What Happens When A Dialysis Bag Containing Pure Water Is Suspended In A Beaker Of Seawater What Would Happen If The Dialysis Membrane Were Permeable To Water But Not Solutes
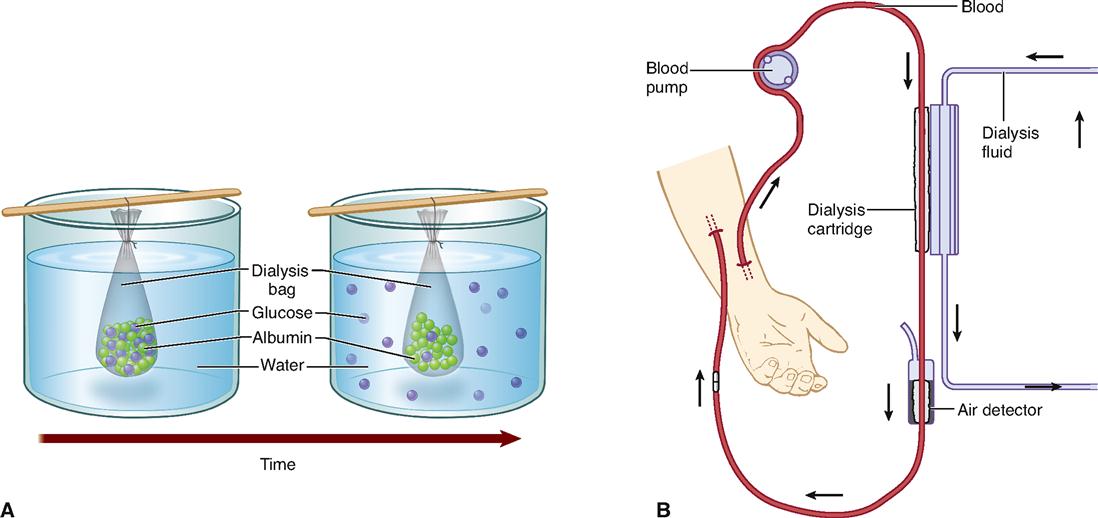
Physiology Of Cells Basicmedical Key

Solved Structure Com Co In Water For 1 Soak Two Lengths Of Chegg Com

Kidney Crew Dialysis Nurse Nephrology Kidney Squad Tote Bag Zazzle Com Tote Bag Casual Accessories Dialysis Nurse

Direct Preparation Of Dialysate From Tap Water Via Osmotic Dilution Sciencedirect

Johns Hopkins Team Develops New Method To Make Kidney Dialysis Fluid For Patients With Covid 19 Johns Hopkins Biomedical Engineering

Osmosis And Diffusion Worksheet Osmosis Teaching Biology Life Science Activities

Cell Membrane And Transport Hw Dr Weiner Ap

Very Simple Diffusion And Osmosis Experiment Osmosis Experiment Teaching Science Biology Labs
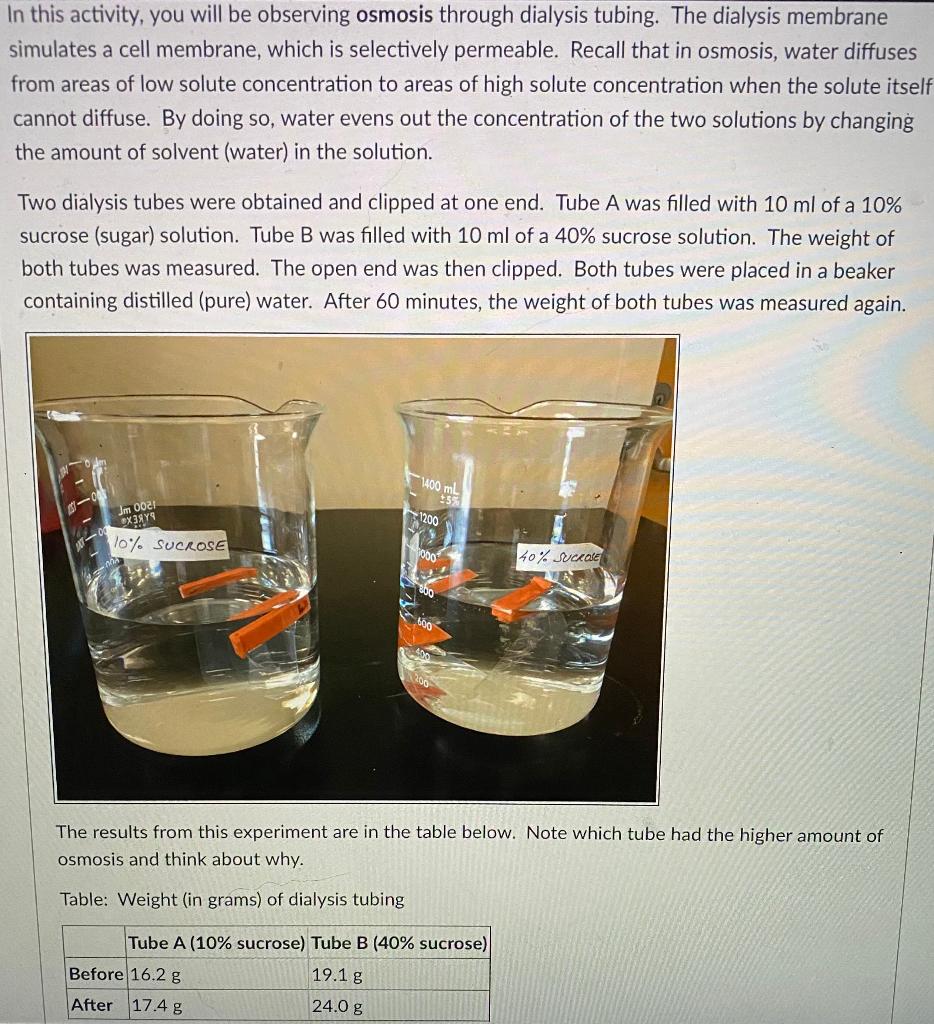
Solved 1 What Happened To The Weight Of Your Dialysis Tubing Chegg Com

Simple Water Experiments To Explain Osmosis And Diffusion Biology Science Fair Projects Science Teaching Resources Science Cells

Rainbow Roses Tote Bag Zazzle Com Rose Tote Bag Rainbow Roses Tote Bag

Comparative Composition Of Dialysis Fluid And Plasma Download Table

How To Do A Kidney Cleansing Fast Healthy Eating Healthy Snacks Healthy Recipes



Comments
Post a Comment